 That is, the number of electrons in indium is forty-nine. Indium atom electron configuration (Bohr model) The atomic number is the number of electrons in that element. 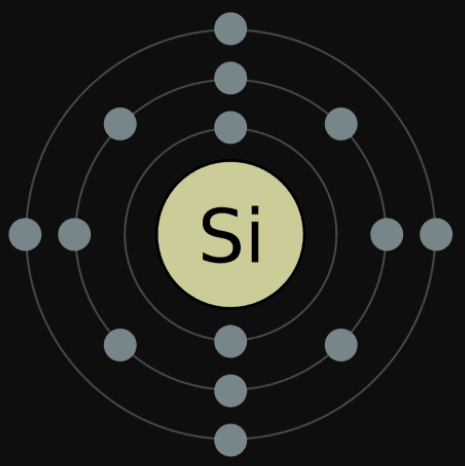
Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons. The maximum electrons holding capacity in N orbit is 2n 2 = 2 × 4 2 = 32. The maximum electrons holding capacity in M orbit is 2n 2 = 2 × 3 2 = 18. The maximum electron holding capacity in L orbit is 2n 2 = 2 × 2 2 = 8. The maximum electron holding capacity in K orbit is 2n 2 = 2 × 1 2 = 2. 
The electron holding capacity of each orbit is 2n 2. K is the name of the first orbit, L is the second, M is the third, and N is the name of the fourth orbit. These circular paths are called orbit(shell). The electrons of the atom revolve around the nucleus in a certain circular path. The complete idea of the orbit is given there. Scientist Niels Bohr was the first to give an idea of the atom’s orbit. 
Indium atom electron configuration through orbit For example Aufbau principle, Hund’s principle, and Pauli’s exclusion principle. Electron configuration through orbital (Aufbau principle)Įlectron configuration through orbitals follows different principles.Electron configuration through orbit (Bohr principle). 
Electron configuration can be done in two ways. The electron configuration of indium is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 1, if the electron arrangement is through orbitals. The arrangement of electrons in indium in specific rules in different orbits and orbitals is called the electron configuration of indium. These electrons are arranged according to specific rules in different orbitals. The total number of electrons in indium is forty-nine. What is the electron configuration of indium? In this article, I have discussed in detail how to easily write the complete electron configuration of indium. Indium is a post-transition metal element. Trending Questions How is temperature affect the spontaneity of a reaction with positive H and S? What is the difference between a endothermic reaction and a exothermic reaction? How do you make 1m kh2po4 buffer? Why is the sodium flame is not visible through the cobalt glass? Total number of electrons in 3d orbital of Cr3 plus is? Hydrogen peroxide peracetic acid acetic acid can these three be shipped in the same box and what happens if they mix? The particle NOT found in the nucleus of the atom is what? What are kinds of acids and their uses? Does scotch contain sodium? How many different elements make up beryllium sulfite BeSO3? Mass number of magnesium? How many moles of carbon are quinine? Is an antacid tablet dissolving in a glass of water chemical change? What is the valency of MnO? Where in Toronto can one purchase low acid coffee? Is a metal surface being ground a chemical change? What is an example of production of gas in a chemical change? What is the distinguishing test between primay secondary and tertiary amine? Which of the following elements exists as diatomic molecules? How much energy does it take to boil 100 mL of water (Refer to table of constants for water.Indium is the 49th element in the periodic table and its symbol is ‘In’. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
